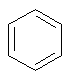
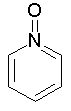
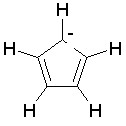
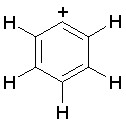
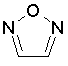
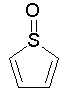
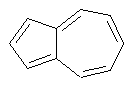
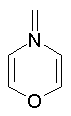
The dProperties algorithm identifies aromaticity regardless of the aromatic bonds defined in the molecule input file. In effect, the user can enter a Kekulè-like structure with alternating single and double bonds or a structure as aromatic directly. dProperties can always recognize aromatic rings only on the basis of atom valences.
A ring is identified as aromatic if it is planar (all atoms are sp2 hybridized), doesn't form exocyclic double bonds and satisfies the Huckel's 4N+2 rule. Moreover, the only allowed heteroatoms are N, O, S, Se, and P.
Rules for counting shared pi electrons:
| 1. | sp2 hybridized neutral C shares one pi electron as in benzene; |
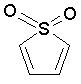
| 2. | O and S share two pi electrons as in furane and thiophene; S in sulfone is also assumed to share a lone pair; |
| 3. | sp2 hybridized N shares one pi electron as in pyridine; |
| 4. | sp3 hybridized N, forming three single bonds, shares two pi electrons as in pyrrole; |
| 5. | sp3 hybridized N, forming two single bonds and one double bond, shares one pi electron if the double bond is not exocyclic, otherwise, the number of shared pi electrons would be zero; |
| 6. | sp3 hybridized N, forming one single and two double bonds, doesn't share pi electrons, excluding N as in pyridine N-oxide which shares one pi electron; |
| 7. | P is treated like nitrogen; |
| 8. | Se is treated like oxygen. |
For charged structures the total number of shared pi electrons is reduced by the positive in-ring charge and increased by the negative in-ring charge.
If the molecule structure is entered by SMILES notation, the dProperties algorithm identifies pyrrolyl N (sharing 2 pi electrons) by the atomic symbol n (or N) and pyridyl N (sharing 1 pi electron) by the symbol n+ (or N+). Therefore, SMILES users are strongly suggested to always specify pyridyl type nitrogen in order to allow dProperties to correctly represent the molecule structure.
If two bonds formed by a sp3 N are originally marked as aromatic, this N is by default identified as pyridyl type. However, if by this assumption the structure does not result aromatic, then the pyrrolyl N hypothesis will be tested.
If a structure exists in tautomeric forms and the user has entered the nonaromatic one, dProperties algorithm cannot recognize it as aromatic.
Note that the dProperties algorithm for aromaticity detection doesn't pretend to account for real molecule aromaticity. It is based on some rigorous rules which have been implemented to recognize most of the common aromatic structures.
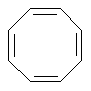
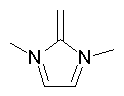
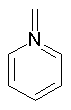
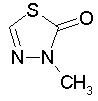
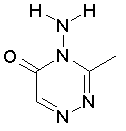
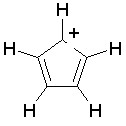
Some examples of aromatic and nonaromatic structures as recognized by dProperties are reported below.
Examples of aromatic structures:
|
|
|
|
|
|
|
|
|
Examples of nonaromatic structures:
|
|
|
|
|
|
|
|
|